
Recipient of NSF SBIR Award

EVAR Therapy Failure
Requires Repeat Intervention.
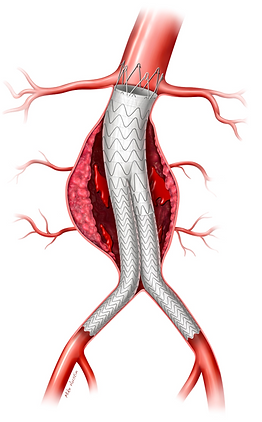
EVAR is the Standard of Care for Abdominal Aortic Aneurysm (AAA) repair.
However, mid to long term outcomes are concerning
1
~1 in 3
EVAR patients undergoes a reintervention
55%
of secondary interventions due to blood flow in the aneurysm sac from type II endoleaks
2
Patients with Type II endoleaks
do far worse than those that do not.
3
5x
more risk of
aneurysm related mortality
8x
more risk of
Aneurysm rupture
10x
more risk of
sac enlargement
20x
more risk of
reintervention
Life Seal Vascular's CYGNUM
Aneurysm Sac Management Device
TM
Simple to Use.

Deployable in 5 mins
No imaging interference
Predictable in location and fit
Only a few SKUs to treat multiple anatomies


Cost Efficient.
Widely Applicable.
Compatible with all EVAR devices
Can be deployed in a wide range of patients and different anatomies
Platform technology for applications in abdominal, iliac, venous and thoracic indications
1. Columbo et. al. Ann Surg 2021; 274: 179
2. Fairman et. al. J Vasc. Surg 2018; 67: 1082
3. Seike et. al. Circulation 2022; 145: 1056
